As Pfizer Side Effects and Medication Safety Standards take the spotlight, this introductory passage invites readers with a blend of informative insights and captivating details, promising a stimulating read that is both educational and engaging.
The subsequent paragraph will delve into the intricacies of the topic, providing a comprehensive overview that sheds light on important aspects of Pfizer vaccines and medication safety standards.
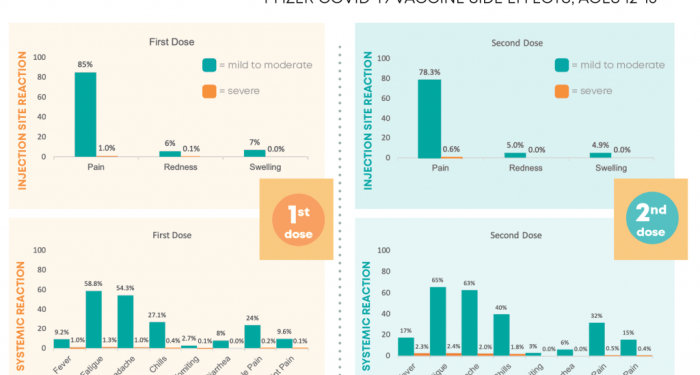
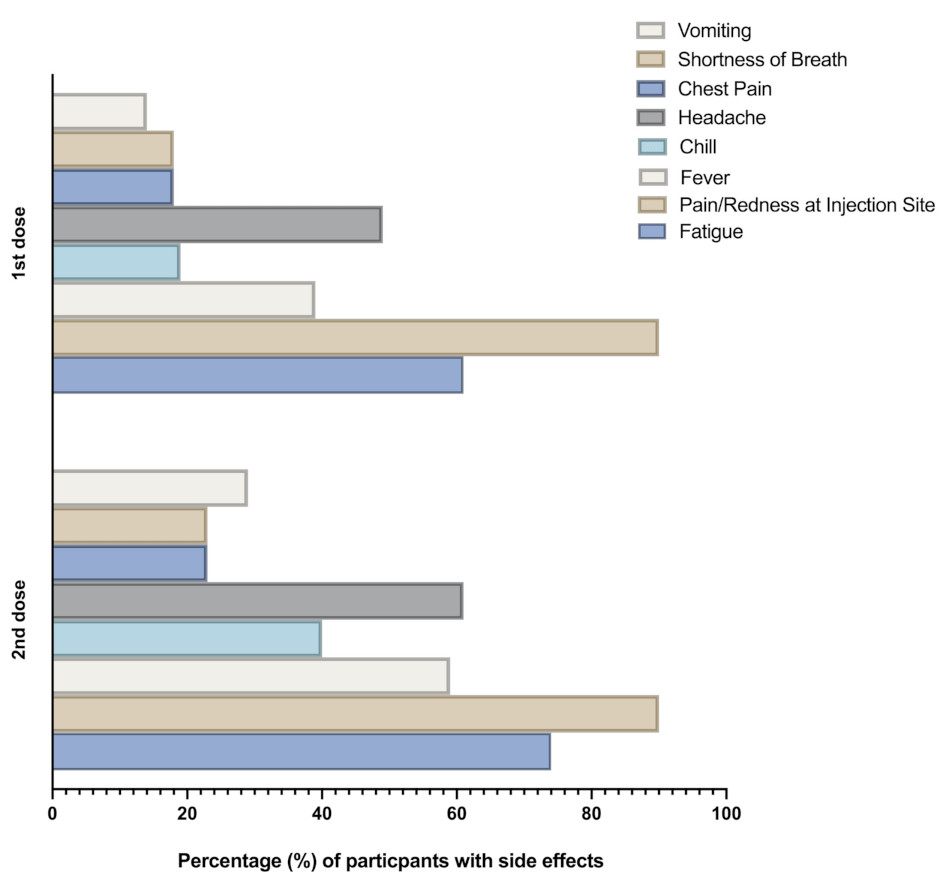
Pfizer Side Effects
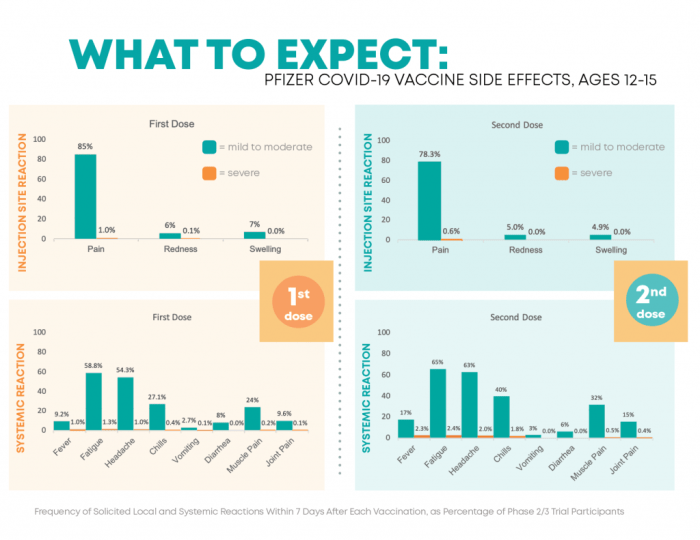
The Pfizer vaccines, like any other vaccine, may cause side effects in some individuals. These side effects are typically mild and resolve on their own within a few days. However, it is essential to be aware of the potential side effects associated with Pfizer vaccines.
Common Side Effects
- Injection site pain, redness, or swelling
- Fatigue
- Headache
- Muscle pain
- Chills
- Fever
Rare but Severe Side Effects
- Myocarditis or pericarditis: Rare cases of inflammation of the heart or its lining have been reported, primarily in male adolescents and young adults within a few days after vaccination.
- Guillain-Barré Syndrome: An extremely rare neurological disorder that causes muscle weakness and paralysis, with a few reported cases following Pfizer vaccination.
- Thrombosis with thrombocytopenia syndrome (TTS): A rare blood clotting disorder associated with low platelet counts, observed in some individuals after vaccination.
Comparison with Other COVID-19 Vaccines
When comparing the safety profile of Pfizer vaccines with other COVID-19 vaccines, Pfizer has shown similar side effect profiles to Moderna, another mRNA vaccine. However, the occurrence of rare but severe side effects may vary between different vaccines. It is essential to consult healthcare professionals for personalized advice on vaccine selection based on individual health conditions and risk factors.
Medication Safety Standards
The development of Pfizer vaccines adheres to stringent safety protocols to ensure the efficacy and safety of the medication
Monitoring and Reporting Systems
- Continuous monitoring of vaccine safety data post-approval is conducted through various surveillance systems.
- Adverse events are reported and investigated promptly to assess any potential risks associated with the vaccine.
- Patients are encouraged to report any side effects experienced after vaccination for further evaluation.
Regulatory Bodies
- The Food and Drug Administration (FDA) plays a crucial role in evaluating and approving Pfizer vaccines based on safety and efficacy data.
- The Centers for Disease Control and Prevention (CDC) provides guidelines and recommendations for the safe administration of vaccines.
- The World Health Organization (WHO) collaborates with regulatory agencies globally to ensure medication safety standards are met across borders.
Last Word
In conclusion, this discussion on Pfizer Side Effects and Medication Safety Standards encapsulates the key points discussed, offering a concise yet thorough wrap-up that underscores the significance of vaccine safety and monitoring protocols.
Popular Questions
What are some common side effects of Pfizer vaccines?
Common side effects include soreness at the injection site, fatigue, and mild fever, which usually subside within a few days.
How do Pfizer vaccines compare in terms of safety with other COVID-19 vaccines?
Pfizer vaccines have demonstrated a high safety profile, comparable to other leading COVID-19 vaccines, with rare severe side effects reported.
Which regulatory bodies oversee medication safety standards for Pfizer vaccines?
Regulatory bodies such as the FDA and EMA play crucial roles in ensuring the safety and efficacy of Pfizer vaccines through rigorous evaluation processes.